The European Union is working on a proposal to ban PFAS due to their harmful environmental impact. Under the REACH regulation, a complete ban on PFAS is proposed for 2025.
However, Annex XVII includes an exemption for category III PPE (Personal Protective Equipment) clothing, allowing the use of PFAS until 2037 because of their protective properties. Category III includes the standards EN13034:2005 + A1:2009, EN11612, and EN61482.
What is PFAS?
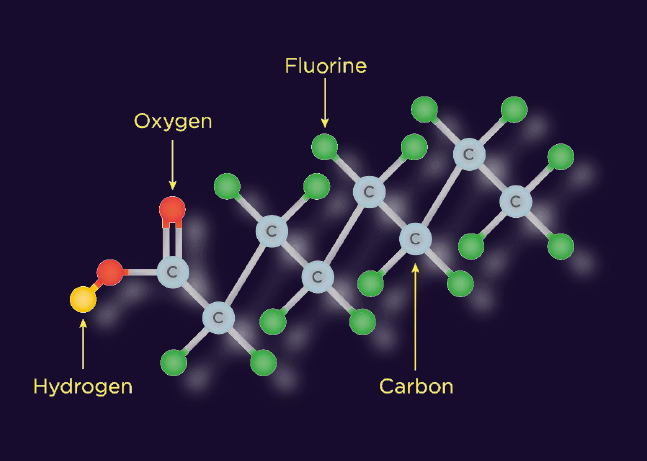
PFAS stands for poly- and perfluoroalkyl substances – a group of man-made chemicals known for their water-, oil-, dirt-, and chemical-repellent properties. PFAS are widely used in everyday products such as cookware, clothing, cosmetics, packaging, and firefighting foams.
Alarmingly, PFAS have even been found in food products like eggs, and in the blood of polar bears in the Arctic. In textiles, PFAS are used in finishes and membranes, such as fluorocarbon coatings and EPTFE membranes, to enhance resistance to water, oil, and chemicals.
But is it truly essential to remain dependent on these "forever chemicals"?
Back to the basics
The principle of liquid repellency is based on surface tension. Surface tension results from the tendency of molecules within a substance to attract each other rather than adhere to another surface. A well-known example is that oil and water don’t mix due to their different surface tensions.
Repellent finishes increase the surface tension of the fabric, making it more resistant to liquids. Every liquid has a measurable surface tension, expressed in mN/m. For example, water has a surface tension of 72 mN/m, while olive oil measures around 32 mN/m.
These values help determine the level of repellency needed for a fabric. Below is a scale of the most commonly used liquids to assess the need for repellency.
PFAS and surface tension
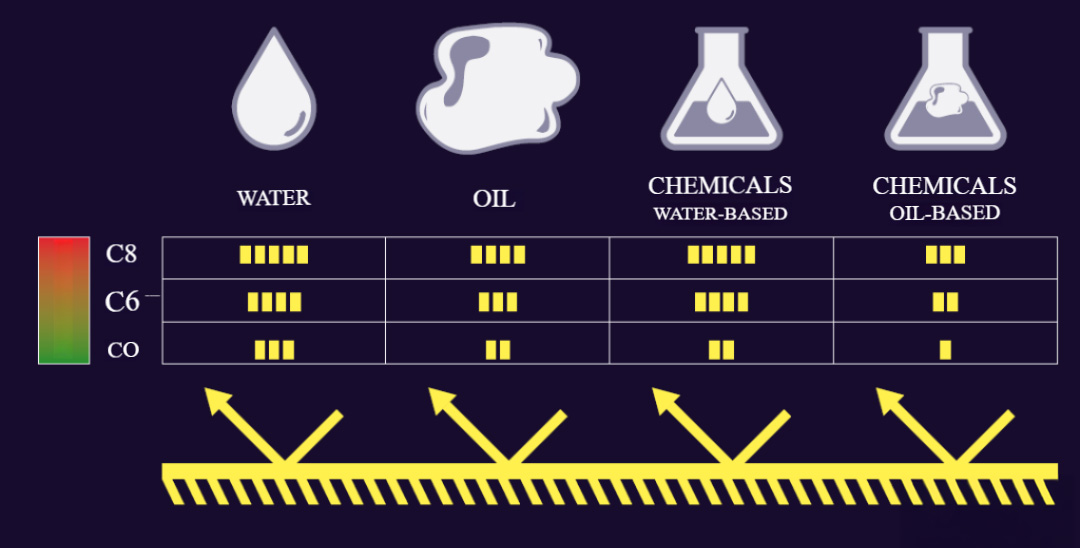
A fluorocarbon finish increases the surface tension of the fabric, improving resistance to water, oil, and chemicals. So far, no other chemical substance is known to achieve the same level of surface tension.
But this is black-and-white thinking. At this point, it’s important to distinguish between water and oil, as well as the chemicals based on them. As shown on the scale below, water has a much higher surface tension – which means PFAS isn’t needed for water repellency. For oil, and therefore oil-based chemicals, which have a lower surface tension, PFAS is still required.
It's all in the nuance
So, are you fully protected? The PFAS-free solution, the C0 finish, also offers good water repellency and therefore protection against water-based chemicals.
Let’s be clear: EN13034 includes a test method (the gutter test) based on liquid droplets – 10 ml over a period of 10 seconds (at a 45-degree angle). To visualize this: that’s about two teaspoons of liquid.
This standard defines a minimum level of protection against chemicals. A PFAS-free solution also meets this minimum protection level when it comes to water-based chemicals, as required by EN13034.
Looking ahead
To highlight HAVEP’s position on PFAS, a distinction will be made between water- and oil-based chemicals, referred to as class 1 and class 2.
Regarding NPDs: all garments that are not EN13034-certified and do not require oil (or oil-based chemical) repellency will be switched to PFAS-free finishes.
For category III PPE garments, the key question is whether oil resistance is necessary. While sustainability is vital, safety always comes first.
Back to the customer
The decision to use PFAS isn’t black and white – it depends on the needs of your customer and the chemicals they work with. By understanding those needs, you can match the right garment with the appropriate level of protection.
Not sure what your customer needs? Let’s figure it out together!
| Product | Chemical formula | Surface tension (mN/m) |
|---|---|---|
| Pentane | C₅H₁₂ | 16.0 |
| n-Hexane | C₆H₁₄ | 18.4 |
| n-Heptane | C₇H₁₆ | 20.1 |
| Octane | C₈H₁₈ | 21.8 |
| Isopropyl alcohol 70% | (CH₃)₂CHOH | 22.0 |
| Methanol | CH₃OH | 22.6 |
| Ethanol | C₂H₆O | 22.3 |
| Acetone | C₃H₆O | 23.7 |
| n-Butanol (class 2) | C₄H₁₀O | 24.6 |
| Diesel | — | 25.0 |
| n-Hexadecane | C₁₆H₃₄ | 27.5 |
| 2-Ethoxyethanol | C₄H₁₀O₂ | 28.2 |
| Benzene | C₆H₆ | 28.2 |
| Diethylene glycol | C₄H₁₀O₃ | 30.1 |
| o-Xylene | C₈H₁₀ | 30.6 |
| Toluene | C₇H₈ | 28.5 |
| Tetradecane | C₁₄H₃₀ | 30.8 |
| Olive oil | — | 32.0 |
| Cyclohexanol | C₆H₁₁OH | 32.9 |
| Acetic acid 90% | HAc | 41.0 |
| Acetic acid 50% | HAc | 48.1 |
| Isopropyl alcohol 15% | (CH₃)₂CHOH | 48.2 |
| Sulfuric acid 30% | H₂SO₄ | 55.0 |
| Blood | — | 55.9 |
| Acetic acid 20% | HAc | 58.5 |
| Hydrochloric acid 45% | HCl | 60.9 |
| Nitric acid 50% | HNO₃ | 61.0 |
| Glycerol | C₃H₈O₃ | 63.0 |
| Sulfuric acid 96% | H₂SO₄ | 63.0 |
| Hydrochloric acid 25% | HCl | 65.8 |
| Water | H₂O | 72.8 |
| Caustic soda 10% | NaOH | 75.0 |
| Caustic soda 50% | NaOH | 85.0 |
Still have questions? Feel free to contact us!
